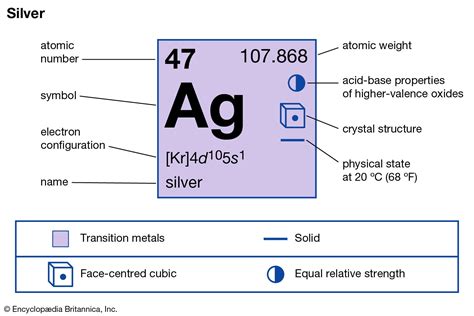
Understanding the significance of the silver symbol in chemistry is essential for anyone studying the foundational elements of the periodic table. Silver, known in Latin as “argentum,” holds a unique position in the realm of chemistry due to its extensive use in jewelry, electronics, and its remarkable antimicrobial properties. Delving into its chemical identity, its symbol ‘Ag’ reveals much about its historical and scientific relevance.
Key insights box:
Key Insights
- The silver symbol ‘Ag’ reflects both its Latin name ‘argentum’ and its extensive historical use.
- Silver’s high electrical and thermal conductivity make it vital in modern electronics and conductive applications.
- Consider silver’s role in catalytic reactions due to its unique properties, for practical applications in various industries.
Silver’s Symbol ‘Ag’: Historical and Chemical Background In chemistry, the symbol ‘Ag’ for silver signifies a rich historical and scientific heritage. The Latin name ‘argentum’ translates directly to’silver’ in English, which is why its symbol is derived from the first two letters of the word. Historically, silver has been highly prized for its aesthetic value and versatility. The ancient civilizations, including the Egyptians and Romans, utilized silver extensively for coinage and art. Today, its symbol continues to play a critical role in modern science, as silver’s unique properties drive numerous technological advancements.
Silver’s Practical Applications in Modern Chemistry The unique properties of silver, such as its excellent conductivity and unique antimicrobial characteristics, make it indispensable in various modern applications. Its high electrical and thermal conductivity are particularly crucial in the electronics industry. Silver is the best conductor of electricity among metals, making it essential in circuits, microprocessors, and solar cells. Furthermore, its antimicrobial properties have driven its use in medical devices, coatings, and textiles where its ability to inhibit microbial growth is beneficial. In catalysis, silver is used in reactions such as hydrogenation and ethylene oxidation due to its ability to facilitate chemical transformations efficiently.
Frequently Asked Questions (FAQ)
What are the primary industrial uses of silver today?
Today, silver is primarily used in the electronics industry for its superior conductivity, in medical applications for its antimicrobial properties, and in photography due to its exceptional light sensitivity. Additionally, silver plays a significant role in catalytic reactions and is utilized in high-quality jewelry and silverware.
How does silver compare to other metals in terms of conductivity?
Silver has the highest electrical conductivity of all metals, making it superior to copper and gold in this regard. While gold is valued for its resistance to corrosion, silver’s unmatched conductivity makes it irreplaceable in certain high-demand technological applications.
In conclusion, the chemical symbol ‘Ag’ for silver not only encapsulates its Latin origin but also underscores its wide-ranging applicability in modern science and industry. Its exceptional properties have kept it at the forefront of technological and medical advancements, highlighting the enduring importance of this precious metal in contemporary applications.